 Energy is absorbed because the negative ion is gaining a negative electron. The second electron affinity is endothermic and is defined as the energy absorbed when one mole of electrons is gained by a -1 ion. For example oxygen andsulfur form -2 ions and have a first and second electron affinity. NOTE: Non-metal elements with -2 ions have two electron affinities. Exothermic because the gaseous atom needs to release energy in order to slow down sufficiently to attract and an electron. The energy released when one mole of gaseous chlorine atoms gains one mole of electrons to form a -1 charged ion. Endothermic because the bonds between the Cl-Cl atoms are being broken.Ĭ) ∆H2° standard enthalpy of first electron affinity of chlorine. The energy required to change one mole of gaseous chlorine molecules into one mole of gaseous chlorine atoms. They are exothermic because new bonds are being made between the atoms of the elements sodium and chlorine.ī) ∆H1° standard enthalpy of atomization of chlorine. Mole of a NaCl is formed from its elements under standard conditions. Example in calculation of lattice enthalpy of NaCl as below:Ī) ∆Hf° standard enthalpy of formation of sodium chloride. Born-Haber Cycles (named after Fritz Haber and Max Born) are simply energy / enthalpy cycles that show how ionic compounds are formed from their constituent elements.ĥ. Remember that bond breaking is endothermic and bond making is exothermic.Ĥ. Because all the bonds in the ionic lattice are made, it is an exothermic process, ∆H is negative.'ģ. Because all the bonds in the ionic lattice are broken, it is an endothermic process, ∆H is positive.ī) The enthalpy change that occurs when one mole of a solid ionic crystal is formed form its ions in the gaseous state, at standard temperature and pressure. Ions in the gaseous state, at standard temperature and pressure. Lattice enthalpy is also defined as either the:a) The enthalpy change that occurs when one mole of a solid ionic crystal is broken into its Definition of lattice energy – The enthalpy of formation of 1 mole of a compound from its gaseous ions under standard conditions ((298K, 1atm )Ģ. Exothermic because the gaseous atom needs to release energy in order to slow down sufficiently to attract and an electron.ĬHEMICAL ENERGETICS (cont)7.3 BORN HABER CYCLEħ.3.1 Define lattice energy for simple ionic crystals in terms of the change from gaseous ions to solid lattice7.3.2 Explain qualitatively the effects of ionic charge and ionic radius on the numerical magnitude of lattice energy values 7.3.3 Construct Born-Haber cycle for the formation of simple ionic crystals.ġ. The energy ° released when one mole of gaseous chlorine atoms gains one mole of electrons to form a -1 charged ion. c) H ∆ 2 standard enthalpy of first electron affinity of chlorine. 
Endothermic because the bonds between the Cl-Cl atoms are being broken. The energy required ° to change one mole of gaseous chlorine molecules into one mole of gaseous chlorine atoms. b) H ∆ 1 standard enthalpy of atomization of chlorine. They are exothermic because new bonds are being made between the atoms of the elements sodium and chlorine. The enthalpy change when one mole of a NaCl is formed from its elements under standard conditions. 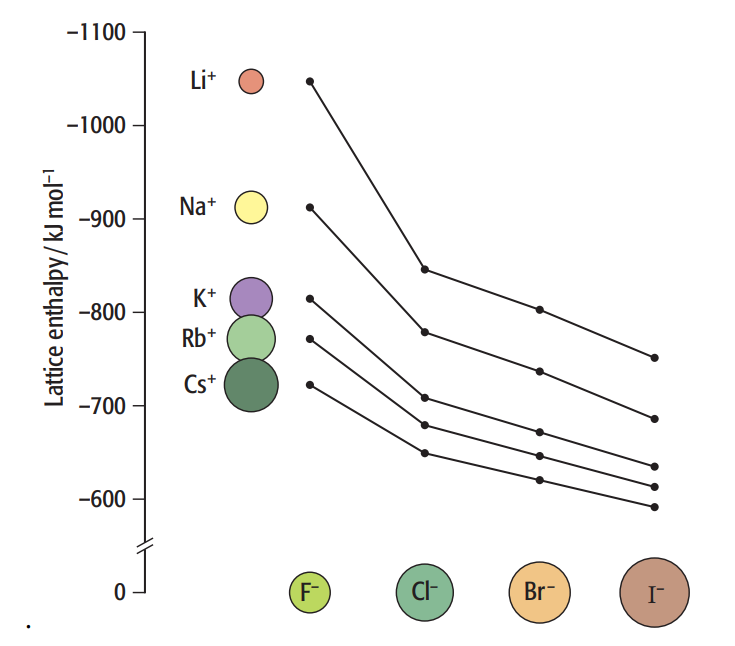
Example in calculation of lattice enthalpy of NaCl as below : a) H ∆ f ° standard enthalpy of formation of sodium chloride. Born-Haber Cycles (named after Fritz Haber and Max Born) are simply energy / enthalpy cycles that show how ionic compounds are formed from their constituent elements. Remember that bond breaking is endothermic and bond making is exothermic. Because all the bonds in the ionic lattice are made, it is an exothermic process, H is negative.' ∆ M + (g) + X - (g) MX (s) 3. ∆ MX (s) M + (g) + X - (g) b) The enthalpy change that occurs when one mole of a solid ionic crystal is formed form its ions in the gaseous state, at standard temperature and pressure. Because all the bonds in the ionic lattice are broken, it is an endothermic process, H is positive. Lattice enthalpy is also defined as either the: a) The enthalpy change that occurs when one mole of a solid ionic crystal is broken into its ions in the gaseous state, at standard temperature and pressure. Definition of lattice energy – The enthalpy of formation of 1 mole of a compound from its gaseous ions under standard conditions ((298K, 1atm ) 2.  
CHAPTER 7 (Module 2) CHEMICAL ENERGETICS (cont) 7.3 BORN HABER CYCLE 7.3.1 Define lattice energy for simple ionic crystals in terms of the change from gaseous ions to solid lattice 7.3.2 Explain qualitatively the effects of ionic charge and ionic radius on the numerical magnitude of lattice energy values 7.3.3 Construct Born-Haber cycle for the formation of simple ionic crystals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
